Low Dosage Gas Hydrate Inhibitor-LDHI
LDHI's Technical Edge Redefines Inhibition Strategies
Low Dosage Gas Hydrate Inhibitor-LDHI
Engineering Precision in Hydrate Inhibition for Enhanced Operational Efficiency
Explore innovative non-thermodynamic inhibitors that diverge from conventional options such as methanol (MeOH) and monoethylene glycol (MEG). Unlike traditional inhibitors, these cutting-edge substances don't alter thermodynamic hydrate equilibrium conditions; instead, they disrupt the hydrate formation process.
Requiring significantly lower dosages compared to thermodynamic inhibitors, these compounds fall into the category of "low dosage hydrate inhibitors" (LDHI), with effective concentrations typically ranging from 500 ppm to 3% of the total water volume treated. This not only minimizes costs but also mitigates storage and handling risks associated with thermodynamic inhibitors.
KHI
Kinetic Hydrate Inhibitors
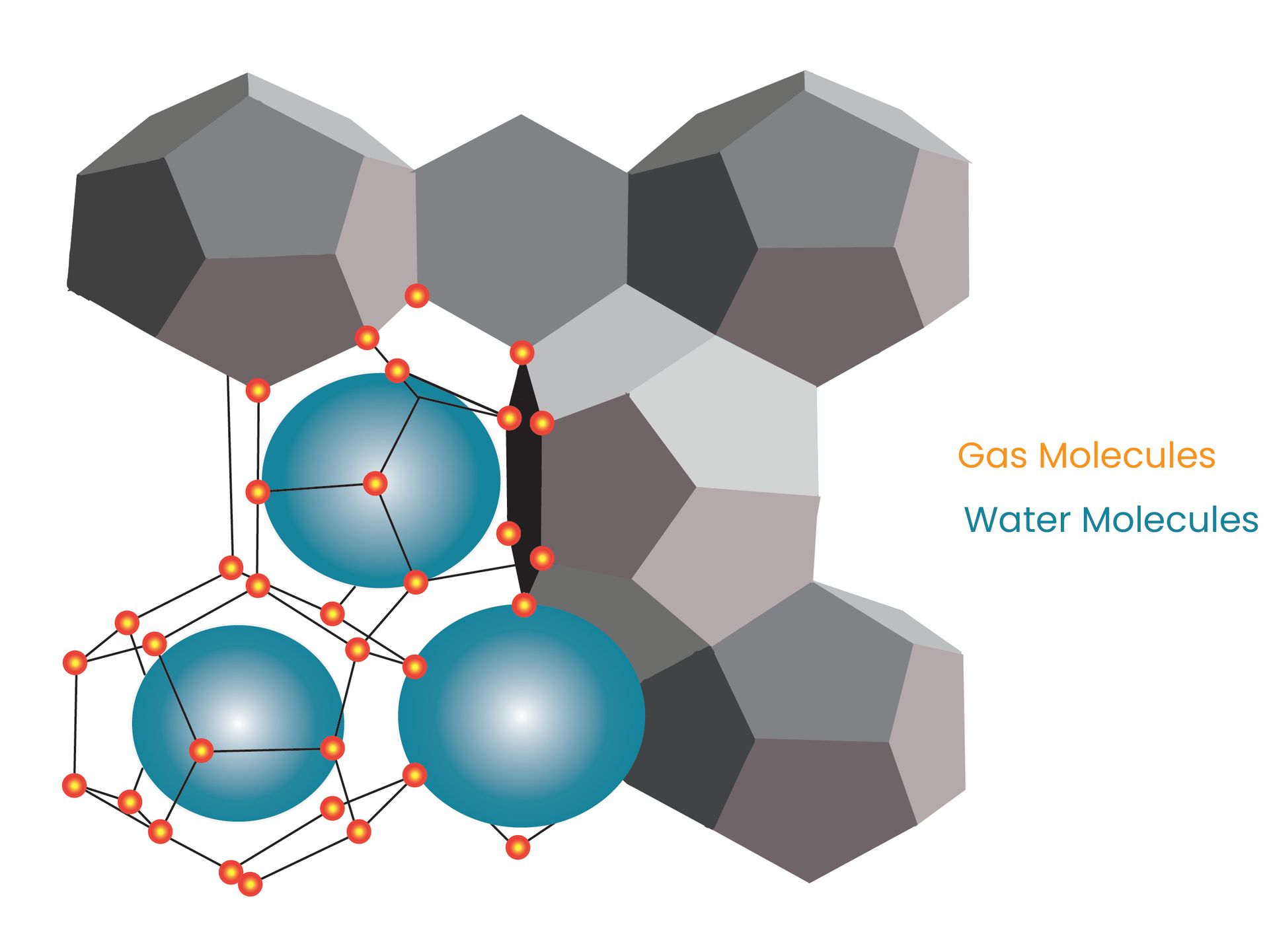
DOSAS low-dosage hydrate inhibitors (LDHI) exhibit effective interference with the nucleation and crystallization of hydrate molecules, thereby prolonging the time required for hydrate formation, known as the induction time. EmendaFlow™ gas hydrate inhibitors, specifically termed kinetic hydrate inhibitors (KHI), predominantly consist of polymer-based materials in aqueous or glycol-based solvent systems. The induction time is influenced by subcooling, representing how much the actual temperature is below the hydrate formation temperature.
AA
Anti-Agglomerant Hydrate Inhibitor
Low-dosage hydrate inhibitors (LDHIs) can effectively modify hydrate particles by either adsorbing onto or integrating into the crystals. To boost anti-agglomerant performance, diverse strategies focus on optimizing the balance between hydrophilic and lipophilic group structures. EmendaFlow™ specialized chemicals are equipped with at least one long hydrocarbon chain, serving to hinder the agglomeration of hydrate particles and ensuring their dispersion as a slurry. Due to this distinctive property, these chemicals are commonly known as anti-agglomerant hydrate inhibitor.
Evaluate LDHI
Gas Hydrate Inhibitors
DOSAS' capability to evaluate hydrate inhibitors using rocking cell equipment provides a clear understanding of hydrate deposits' morphology and distribution. Beyond qualitative visual output, quantitative data can be recorded, including pressure, temperature, and ball travel time within the rocking cell to determine the initiation time of hydrate formation, hydrate volume fraction derived from the pressure trace, and slurry flowability.
Modelling the
Behaviour of Gas Hydrates
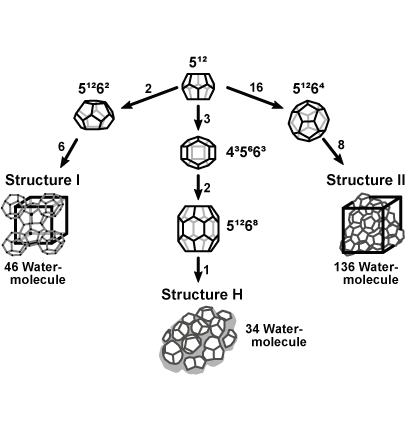
DOSAS assesses production system risks using thermodynamic modelling. Unlike immediate phase boundary crossing, DOSAS employs a kinetic model to identify delays in hydrate formation detection after entering the stable region.